Lumpy Spectra?
I’ve been rabbiting on about “full blackbody spectra” and “lumpy spectra”, perhaps I should show some examples. These are taken from the Lighting Passport.
I show both the spectral curve and the “spider plot” of CRI values, showing how well each light would render each CRI color. In the CRI plots, R1-R8 are colors of mild saturation and brightness, while R9-R15 are strongly saturated (thus more demanding, narrow-band) colors. R9, in particular, corresponds to a saturated red. The Ra value is the average for R1-R8 and is the CRI number normally quoted for a light: note that by excluding the peakier, more saturated colors, CRIs rate lights more favorably than they would if the ratings included the rendering quality of the trickier colors!
Full, Fat-Spectra, Blackbody-like Sources
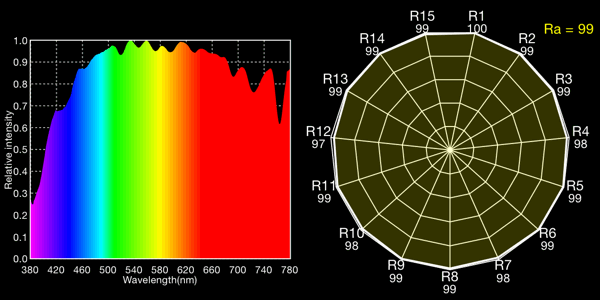
Direct sunlight, 5021K
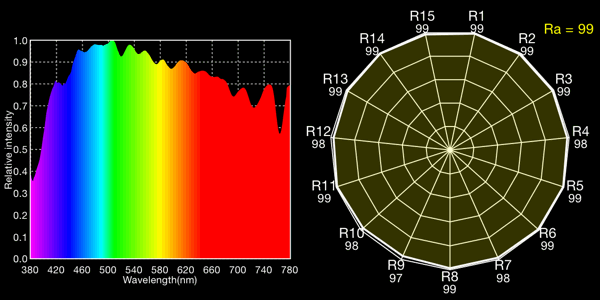
Overcast sky, 5673K
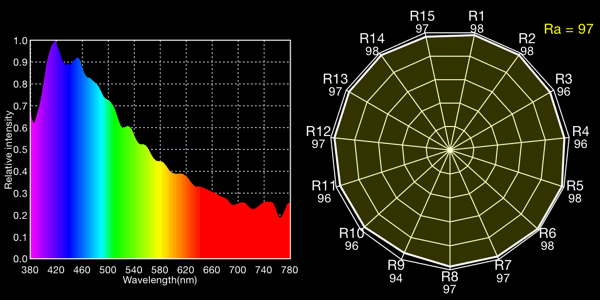
Blue, open sky, 13,759K
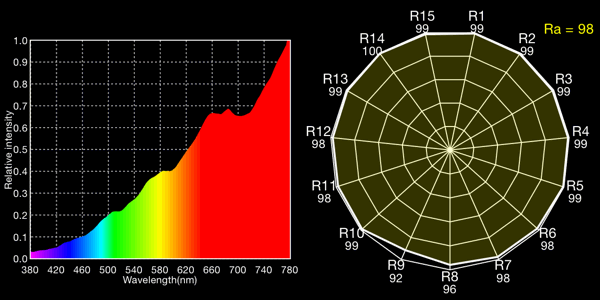
Halogen lamp, 2720K
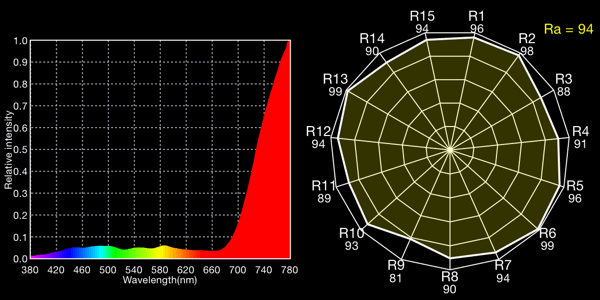
Halogen lamp with 1.5 CTB gels, 5021K
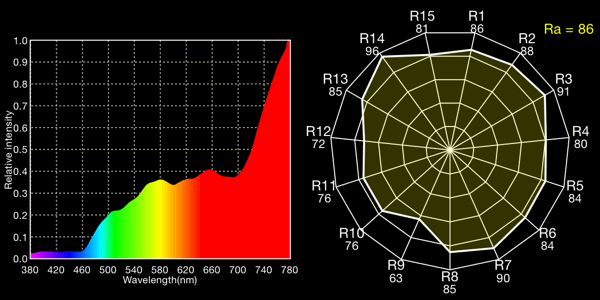
Halogen lamp with CC 30G (plus green), 3147K
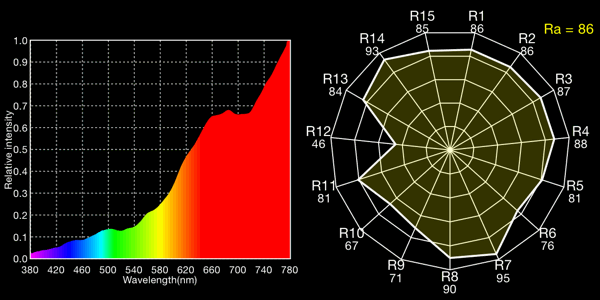
Halogen lamp with CC 30M (minus green), 2132K
Interesting how much near-infrared gets through the gels! Remember that the response curve of most cameras cuts off or diminishes greatly around 700nm, so the big spikes in the right-hand side of the curves won’t register as strongly on-camera as these plots would make you think.
Lumpy-Spectra Sources
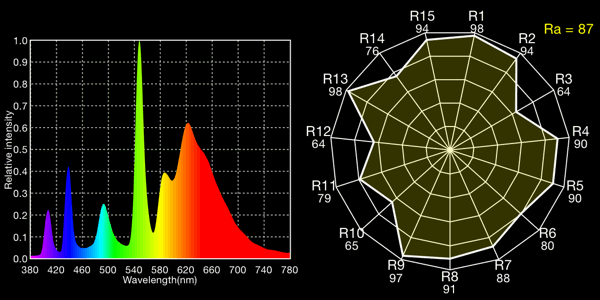
Kino Flo 2900K tubes, 2959K measured
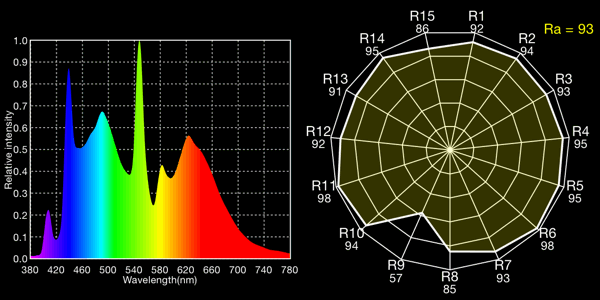
Kino Flo 5600K tubes, 6530K measured (this time, not all times!)
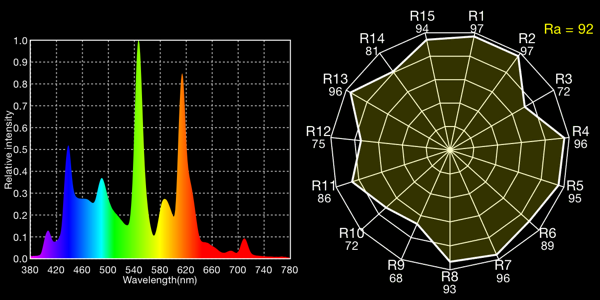
Ott-Lite “natural” fluorescent, 5155K
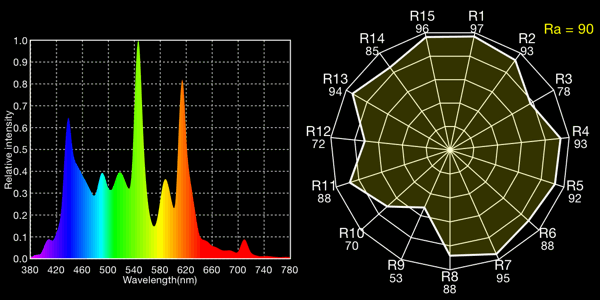
6100K GE household fluorescents, 6098K
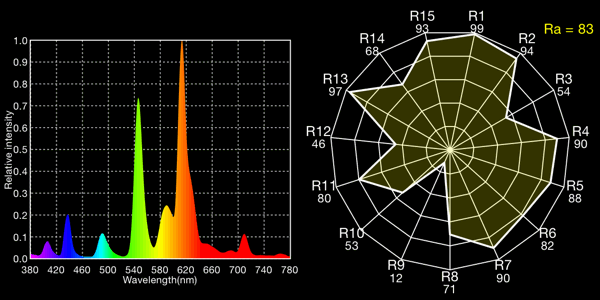
Osram “warm white” compact fluorescent, 2555K
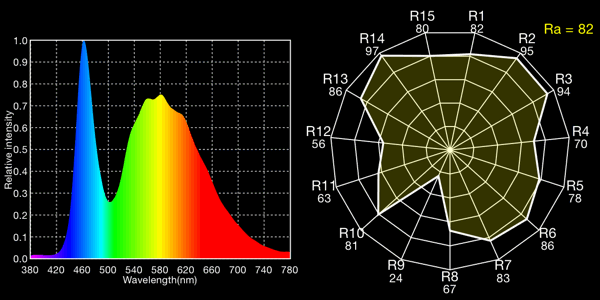
Litepanels Micro 1st-generation, 4442K
All the fluoros show that characteristic “green spike” around 546 to 548nm.
The Litepanels Micro shows a typical phosphor-white LED spectrum, with a fairly smooth curve overall and a large spike at 462nm, presumably from the underlying blue LED. Despite its smooth appearance, it has a poor CRI (not that CRI is the be-all and end-all of lighting quality measurements, but it also has the worst CQS value of all the lights I tested, and I have to admit it does look a bit wonky). Bear in mind that it’s a 1st-generation LED light; recent versions are noticeably better-looking.
One thing occurs to me as I look at these charts.
Art Adams says that all his gaffers, when they use a meter to look at green/magenta correction, use about 1/2 to 2/3 the value the meter suggests. I also found that to be the case; my kitchen fluoros and my desktop fluorescent both read about 30 green, but if I put a 30M gel over the meter, it’ll read about 30 magenta. I really need only a 15M gel to correct a 30G imbalance. Weird.
Art attributes this error to the nature of three-sensor color meters:
The typical flourescent light has huge green and orange spikes, with not a lot else going on. Using a color meter with these sources can result in faulty correction, as colors are typically presented as ratios (orange to blue, green to magenta) and one half of the equation may not be present.
But when it comes to full-spectrum meters like the C-700 or the Lighting Passport, that story won’t work: these meters can see everything in excruciating detail, and should be able to come up with a proper correction. Yet they fall down just as badly as the three-sensor meters in this regard.
I wonder: I did my calibrations under the full, fat spectrum of the halogen light, so the full green and full magenta filters had to filter the entire spectrum. I’m going to guess that the green and magenta gel colors are themselves determined under similar circumstances: the colors are of the density needed to get the +/- 30 tint shift under a nice, predictable full-spectrum source: a 3oM filter has to diminish not only the energy concentrated at the 547nm green spike, but equally strong amounts of cyan-ish and yellow-ish light nearby in the reference lamp’s spectrum.
When a magenta filter is then bunged onto a spiky fluorescent, it’ll step very heavily on the green spike, as it should, but there’s less “fill light” near that green spike in the fluoro’s spectrum – spectral energy that, in the tungsten lamp’s case, fights back against the corrective effect of the magenta gel. With no “supporting cast” of nearby yellow and cyan light alongside the fluorescent’s green spike to push back against the (tungsten-calibrated) magenta gel, the gel is overkill, by as much as a power of two – depending on the selectivity of the filter, and the nearby spectral energy from the light.
I don’t know if that’s the correct story, but it certainly explains the odd disconnect between what the meter says is needed and what the gel actually does.
Lighting Passport and C-700 Compared
The new C-700 and the Lighting Passport both claim to read out spectra in 1nm increments. I put ’em side by side several times to see what they did. The result? They’re close, but not always the same… in the following pix, the C-700 is the one with fewer gridlines on its spectral plot:
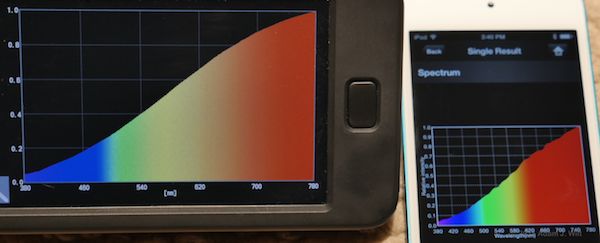
2700K hall lamp: tungsten PAR
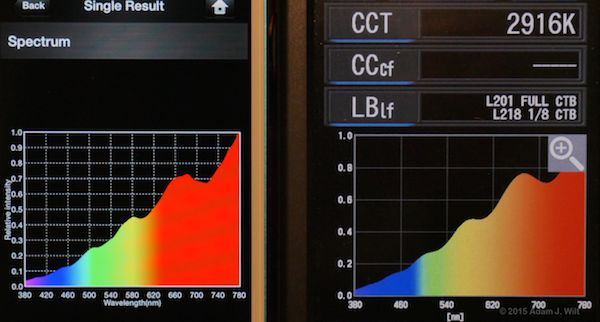
Mole-Richardson 650w Softlight
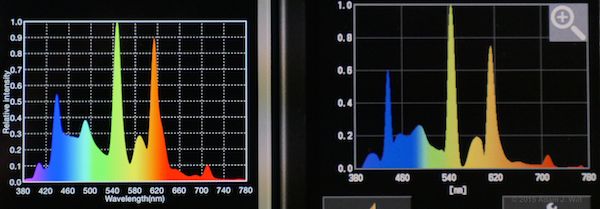
Ott-Lite fluorescent
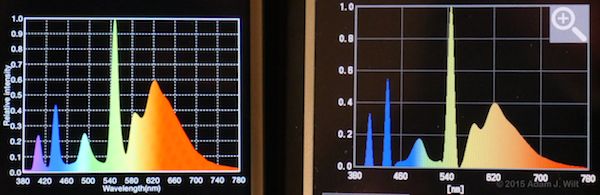
Kino Flo 2900K fluorescents
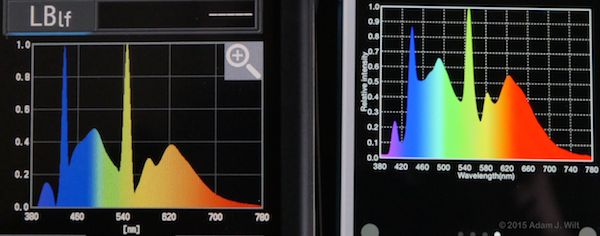
Kino Flo 5600K fluorescents
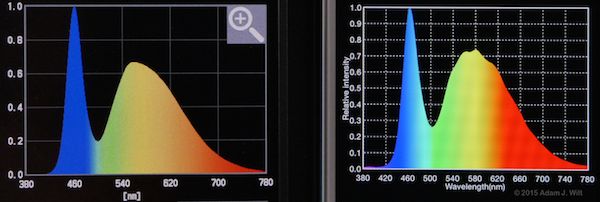
Litepanels Micro LED light
The shapes of the curves generally match, but the peaks and troughs often differ in relative intensities between the two instruments.
The Lighting Passport I have has an actual measurement resolution of 10nm (the fancier one uses 8nm steps); the C-700 claims to “measure in 1 (nm) increments, capturing spikes in light source output, especially fluorescent and LED lighting, providing unmatched color measurement accuracy.” It’s possible the Passport is simply interpolating between those 10nm steps while the C-700 actually measures each nanometer increment directly. [Update 10 March 2015, after talking with Asensetek and getting the final ops manual for the C-700.] The C-700 uses a 128-pixel CMOS sensor, so the best it can possibly resolve is 3.125nm (all the online material I see for the C-700 now claims “1nm output resolution”). Both meters apply some form of interpolation to their measurements; the Asensetek folks claim they’re doing a simple linear interpolation while the C-700’s math is smoothing the output more. So it’s possible the differences in the curves are due both to different sampling intervals and to different interpolation math.
Or it’s possible that the two instruments just have differences of opinion.
If that were the case, it’d be nothing out of the ordinary. When it comes to color metering, it seems like every measurement is subject to differences of opinion!
Disclosure: I bought the Lighting Passport Essence, the used Minolta Color Meter II, and about $50 of color-correction gels with my own funds. The two Sekonics were lent by Art Adams (in return, I bought him a coffee). None of the manufacturers mentioned had any input into my tests nor did they offer any compensation or other considerations for a favorable review.
